
Big Ideas, Real Impact
*
Big Ideas, Real Impact *
Concept to Shelf

End-to-End Private Label Manufacturing — From First Brief to Global Delivery
What began to serve start-ups to craft great products with lower MOQ’s has evolved into something more. We’re proud of where we’ve been and even more excited for what’s ahead. What sets us apart isn’t just our process—it’s the intention behind it. We take time to understand, explore, and create with purpose at every turn.
PHASE 1 - Brief & Quotation
Understand & Propose
We receive your enquiry and arrange a briefing meeting to understand your product concept, target format, commercial objectives, and market. Based on what we hear, we identify the most appropriate development route and prepare a quotation.
Three Development Routes:
Route A — Stock Formula Your concept is matched to a formulation from IGN's catalogue. Samples are prepared and submitted immediately. Fastest route to market.
Route B — Catalogue Adaptation An existing IGN formulation is modified to meet your specific requirements — adjusted actives, fragrance, texture, or format.
Route C — Bespoke R&D A new formulation is developed from scratch by our in-house R&D team. Selected where the brief requires unique performance, proprietary actives, or a format not covered by the existing catalogue.

PHASE 2 - Formulation & Samples
From Agreement to Approved Sample
Once the quotation is agreed and the development route confirmed, formulation begins.
Stock and adaptation routes move to sample preparation immediately. Bespoke R&D begins full development — active selection, delivery system design, texture profiling, and lab batch production.
Lab samples are submitted for your evaluation — texture, application, sensory profile, and performance. Feedback drives iteration until the formulation is approved.
Note: We enable seamless technical transfer of existing formulations to our GMP-certified facilities, ensuring clients move production to us without compromise to sensory profile, stability, or performance.

500 units or 50,000 — the same process. The same rigour.
500 units or 50,000 — the same process. The same rigour.
PHASE 3 - Packaging Development
Technical Selection. Brand Aligned.
Packaging is selected based on formulation compatibility, regulatory requirements, and brand positioning — in that order.
Unbranded samples are submitted for review and selection. Formula–packaging compatibility is tested to ensure durability and product integrity. For brands requiring differentiation, we manage custom mould development and unique component sourcing.
We also support free-issue packaging where customers supply or nominate their own components.

PHASE 4 - Testing & Validation
Nothing Moves to Production Without Passing Validation
Every formulation undergoes a structured testing programme before scale-up is authorised.
Stability testing is conducted over a minimum 12-week accelerated period to assess performance and shelf life under controlled conditions. Microbiological challenge testing confirms preservative efficacy and product safety.
Compatibility testing evaluates the formula–packaging interaction. Where claims require substantiation, performance evaluation and consumer or clinical testing are conducted.

PHASE 5 - Regulatory and Documentation
Multi-Market Compliance. Built In, Not Bolted On.
Regulatory work begins at Phase 1 and is formalised here. Our team prepares all documentation required for your target markets in parallel with development — so compliance is ready when manufacturing is.
EU Cosmetic Regulation 1223/2009 ingredient review and CPNP notification. UK SCPN notification. Product Information File preparation. Safety assessment coordination. Claims substantiation review. Artwork regulatory text verification. GCC and USA market guidance where applicable.

PHASE 6 - Manufacturing & Scale Up
Lab Formula to GMP Production Batch.
Once validated and approved, the formulation moves into GMP-certified production at our facilities. Scale-up is managed by the same technical team that developed the formula — reducing error, maintaining integrity, and ensuring batch-to-batch consistency.
Raw materials are procured and checked on arrival. In-process quality controls are applied at defined checkpoints throughout production. A pre-production sample is submitted for client approval before full batch release.
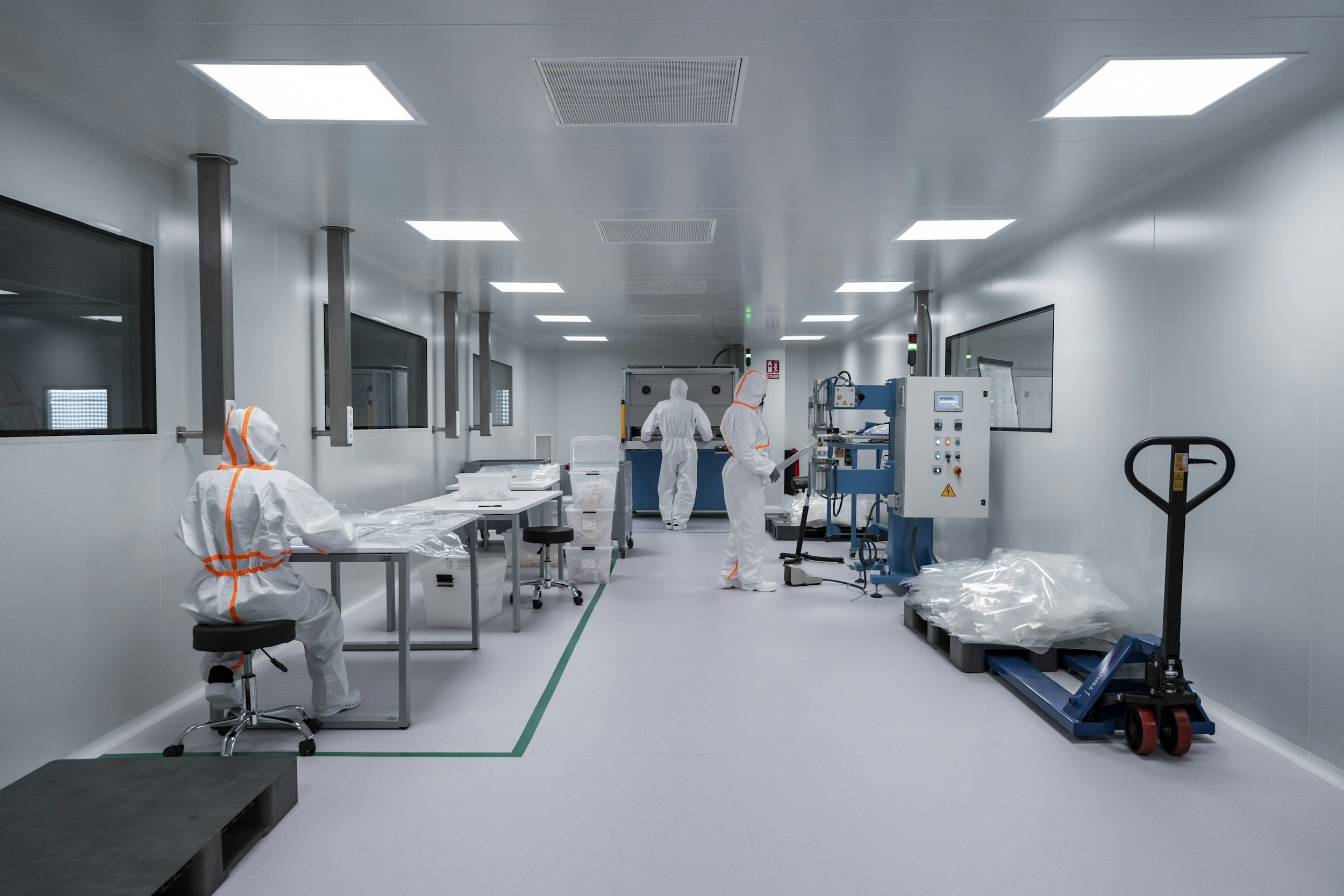
PHASE 7 - Filling & Assembly
Filled. Assembled. Ready for Final Check.
Automated and semi-automated filling lines ensure accurate fill weights, seal integrity, and packaging consistency at every scale. Finished goods are assembled, labelled, and packed.

PHASE 8 - Quality Control and Dispatch
Checked. Certified. Shipped.
Our QA team conducts thorough checks after manufacturing and again after filling — ensuring goods are within specification and fit for purpose at every stage.
Batch documentation and traceability records are completed. A Certificate of Analysis is issued. Final release authorisation is granted and export documentation prepared. International shipment is then coordinated to EU, UK, Middle East and beyond.

Start Your Project
Whether you are a founder launching your first product or an established brand expanding into new markets, IGN provides the manufacturing infrastructure, R&D capability, and regulatory expertise to take your concept to shelf.
Tell us about your project. We will respond within two business days.
